As electrochemical techniques provide a rapid, cheap, and portable means to determine arsenic, it has been widely used. In the literature, various modified electrodes have been developed for the analysis of arsenic, such as As(III)-specific ligands, enzymes, carbon nanotubes (CNTs), graphene, and noble metals (gold silver and platinum), and Au is a superior substrate as the working electrode of the solid metal substrates. Indeed, the construction of gold nanostructures with well controlled morphology on electrodes is not easy to obtain and repeat due to the use of surfactants during the preparation process as well as the surface tension of the bare electrode during modification. It is no doubt that the materials with regular morphology are not difficult to prepare currently. However, a uniform and controllable distribution of prepared nanomaterials on the surface of electrodes are exactly complex. Moreover, the surfactant of nanoparticles will directly affect the performance of electrochemical. In this case, developing an easily constructed electrode with outstanding arsenic detection performance without extensive morphology control is an urgent and important research effort.
Recently, Prof. LIU Jinhuai, 973 chief scientist, and Prof. HUANG Xingjiu from Institute of Intelligent Machines(IIM), Chinese Academy of Sciences (CAS) have a significant progress in designing and constructing a sensing interface for ultra-sensitive detection of As(III) by abundantly dispersing Au nanoparticles on the surface of Fe3O4 nanosphere. The study was published in journal of Analytical Chemistry (http://pubs.acs.org/journal/ancham) with the title of Adsorbent Assisted In Situ Electrocatalysis: An Ultra-Sensitive Detection of As(III) in Water at Fe3O4 Nanosphere Densely Decorated with Au Nanoparticles.
Inspired by the excellent catalytic properties due to the small size of the Au nanoparticles as well as the good adsorption ability of the Fe3O4 nanosphere toward arsenic, researchers report the concept of adsorbent-assisted in situ electrocatalysis for ultra-sensitive detection of As(III) on the Fe3O4 nanosphere densely decorated with Au nanoparticles. The synergistic effects of Au NPs and Fe3O4 successfully and markedly enhance the sensitivity of arsenic detection without morphology control. The redox of As(III) occurs with direct catalysis on the Au NPs surface in situ without diffusing to SPCE after being adsorbed by Fe3O4 nanosphere (Figure).
In addition, according to the theory of diffusion, the inequality d > 2{2D(ΔE/v)}1/2 avoids diffusion zone overlap (known as shielding effects). The inequality  with
with 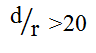 is used, where I2 is the steady-state diffusion-limited current at two interfering coplanar microspheres, and I1 is diffusion-limited current at two independent coplanar microspheres. Keeping d > 20 r is necessary for individual independent diffusion. If not, the adjacent diffusion layers overlap and seriously decrease values of d. When d is far below 20 r, there is complete overlap and a linear concentration profile like the diffusion of a macrodisc. In the system, the mean distance is 2.6 nm versus the mean diameter of 5.4 nm Au particle. It is clear that the separation distance between adjacent Au nanoparticles will cause heavily overlapping diffusion layers and subsequently generate a voltammetric response equivalent to a gold macrodisc electrode as what was found in gold nanoparticle-modified glassy carbon microspheres. Using this new design, good morphology control can be ignored, which is in sharp contrast to the previously developed pure gold nanoparticle-modified electrodes. It is anticipated that it will be possible to design a highly efficient sensing interface to determine heavy metal ions.
is used, where I2 is the steady-state diffusion-limited current at two interfering coplanar microspheres, and I1 is diffusion-limited current at two independent coplanar microspheres. Keeping d > 20 r is necessary for individual independent diffusion. If not, the adjacent diffusion layers overlap and seriously decrease values of d. When d is far below 20 r, there is complete overlap and a linear concentration profile like the diffusion of a macrodisc. In the system, the mean distance is 2.6 nm versus the mean diameter of 5.4 nm Au particle. It is clear that the separation distance between adjacent Au nanoparticles will cause heavily overlapping diffusion layers and subsequently generate a voltammetric response equivalent to a gold macrodisc electrode as what was found in gold nanoparticle-modified glassy carbon microspheres. Using this new design, good morphology control can be ignored, which is in sharp contrast to the previously developed pure gold nanoparticle-modified electrodes. It is anticipated that it will be possible to design a highly efficient sensing interface to determine heavy metal ions.
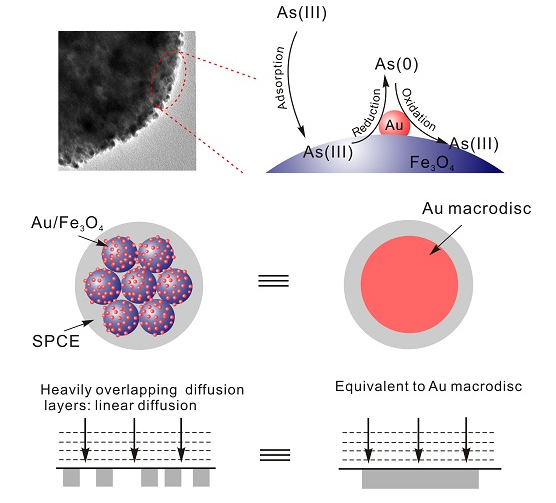
Figure. Schematic of Adsorbent Assisted In Situ Electrocatalysis for As(III) Detection Based on Chemical Architectural Design Taking Advantage of the Synergy between Electrocatalysis of Au NPs and Adsorption of Fe3O4 Nanosphere at Au@Fe3O4-RTIL SPCE. (Image by IIM)
Article link: http://pubs.acs.org/doi/abs/10.1021/acs.analchem.5b02947 (Anal. Chem. 2015. DOI: 10.1021/acs.analchem.5b02947).
Article title: Adsorbent Assisted in Situ Electrocatalysis: An Ultra-Sensitive Detection of As(III) in Water at Fe3O4 Nanosphere Densely Decorated with Au Nanoparticles
Key words: Au; Fe3O4; Electrochemistry; As(III)
Prof. HUANG Xing-Jiu, Ph.D Principal Investigator
Nanomaterials and Environmental Detection Laboratory, Institutes of Intelligent Machines, Chinese Academy of Sciences
(http://www.iim.cas.cn/)
Hefei, Anhui 230031, China
Tel.: +86-551-65591142; fax: +86-551-65592420
E-mail: xingjiuhuang@iim.ac.cn (X.J.H)
